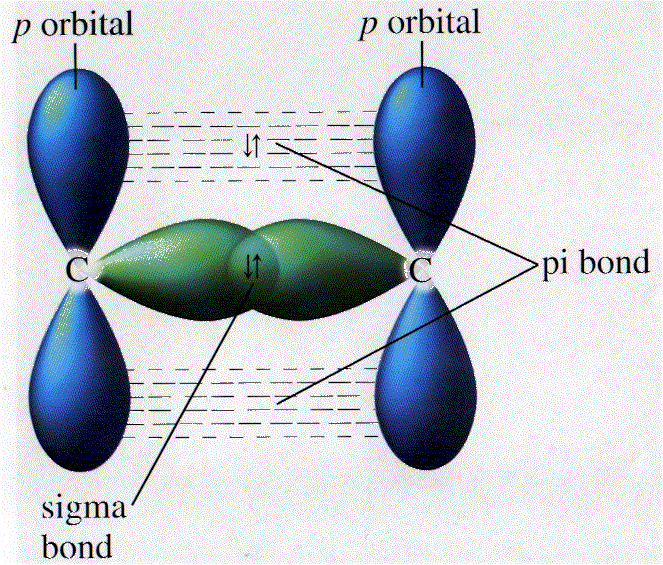
This larger area of overlap corresponds to a higher chance of finding valence electrons between the nuclei of the atoms. Because of the difference in overlap, sigma and pi bonds differ in bonding strength. Strength of Sigma and Pi BondsĪs seen above, sigma bonds have a larger area of bonding overlap. Three types of sigma bonds between s-s, s-p, and p-p atomic orbitals and a pi bond between p-p orbitals. In sigma bonds, head-to-head overlap means that the two orbitals are overlapping directly between the nuclei of the atoms while side-to-side means that the two orbitals are overlapping in a parallel fashion in the space above and below the nuclei. It has absolutely nothing to do with any actual heads but instead this difference refers to where the bonding between orbitals actually occurs. Right, so now you are probably wondering what head-to-head and side-to-side overlap of atomic orbitals even means. Differences between sigma and pi bonds, Source: Tallya Lutfak, StudySmarter originals Must coexist with a sigma bond and found only in double and triple bonds Sigma Bonds (σ)įormed by head to head overlap between atomic orbitals (both hybridized and unhybridized)įormed by side to side overlap between p orbitalsĬan exist independently in single bonds. Check out the explanations for these terms if you need to review them! What Are The Differences Between Sigma and Pi Bonds?īelow is a table highlighting the most important differences you need to know between sigma and pi bonds. To understand Sigma and Pi bonds, you must have a basic understanding of atomic orbitals, hybridization, and hybrid orbitals. When two molecules bond, the orbitals usually combine to form hybrid orbitals such as sp, sp 2, and sp 3. Each of these sets can hold a different amount of orbitals, exist at different energy levels, and have different shapes. There are several types of atomic orbital sets: s, p, d, and f. Remember that covalent bonds form from the overlap of atomic orbitals which are just the space where electrons are likely to be found. Application of Le Chatelier's Principle.Variable Oxidation State of Transition Elements.
Transition Metal Ions in Aqueous Solution.
